
Fact Sheets And Publications
Nitrogen Cycling in Agriculture
April 2025 | Written by: Amy L. Shober
Introduction and Purpose
Manures, organic residuals (e.g., biosolids, food processing wastes), and many commercial fertilizers available to producers contain nitrogen (N). Nitrogen is an important plant nutrient that is often deficient in the sandy soils that dominate Delaware, so application of manures or fertilizers is necessary to produce maximum economic yields. However, if organic residuals or fertilizers are misused, N can be transported from agricultural fields with rain or irrigation water, possibly contaminating nearby water bodies. Understanding how N reacts in the landscape can help us maximize plant growth and crop yields, while minimizing harmful losses of N to the environment. This document helps agricultural producers understand how N acts in the environment through the N cycle to guide maintenance and sustainability of agricultural crop production.
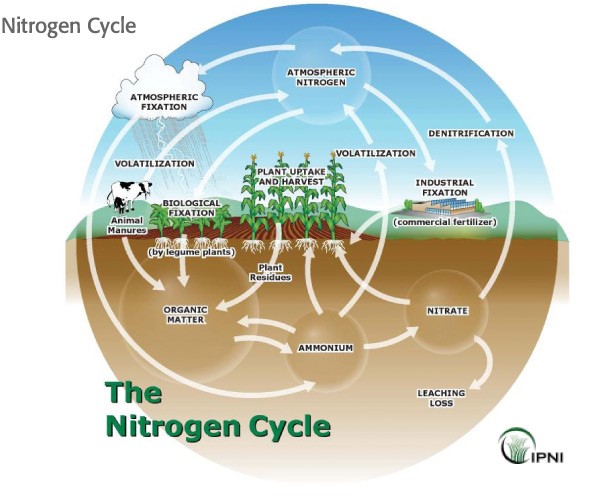
The Nitrogen Cycle
We begin our discussion of the N cycle in a typical grain crop rotation (corn, wheat, double-crop soybean) by considering the plant residue left on the field surface after harvest. Crop residues that are left on the soil surface or incorporated into the topsoil during tillage provide a source of organic matter to the soil. Organic matter contains forms of N that are not available to growing plants. However, soil microorganisms, including bacteria and fungi, change the complex, unavailable forms of N into forms that are plant-available. Inorganic N is then taken up by the roots of the next growing crop in the rotation. The cycle repeats when this crop is harvested (Figure 1). This description is an over-simplified example of the N cycle. Let’s next look at the cycle in more detail.
Sources of Nitrogen
Plant residues are not the only source of N in the soil (Figure 1). The fertilizer industry also manufactures agronomic fertilizers that are used by growers as part of a comprehensive crop fertility program. Most commercial fertilizers contain plant-available N in one of two inorganic forms: ammonium (NH4+) or nitrate (NO3-). Another form of N that can be synthesized by fertilizer manufacturers and is commonly used by growers is urea. Once urea is applied to the soil, it is quickly converted to ammonium by a natural endogenous soil enzyme. Synthetic N fertilizers containing ammonium, nitrate, or urea are considered to be “quick-release” fertilizers that rapidly become plant available. This means that the N contained in the synthetic fertilizers dissolves in water almost immediately after application.
Since dissolved N can be an immediate environmental hazard (more information below), quick-release fertilizers are most effective when applied at relatively low rates (per single application) at times when the crop is actively taking up N. Some companies also produce slow-release or controlled-release fertilizers that are designed to delay the release of plant-available N. Slow- or controlled-release formulations allow for more N to be applied during a single application than when using quick-release fertilizer. It is important to understand that quick-, slow-, and controlled-release N fertilizers can be equally harmful to the environment if they are not applied properly.
Organic soil amendments like manures, composts, or biosolids contain mainly unavailable organic forms of N (i.e., chlorophyll, proteins, etc) and small amounts of plant available inorganic N (primarily ammonium). Organic amendments are often derived from animal wastes or plant residues. Like the crop residues from the earlier example, the organic N in these materials is not plant-available and must be converted to ammonium by soil microbes. Remember, ammonium is the same form of N available in many synthetic fertilizers.
Another source of N to the soil is the atmosphere (Figure 1). The earth’s atmosphere contains 78% N as N2, a non-reactive gas. While most plants and animals cannot use N2, there are a few specialized microbes that can convert this gas into a plant available form of N. These N fixing microbes can live freely in the soil or can colonize the roots of legumes, such as soybean, alfalfa, clovers, lima beans, and peas. Legumes, mainly soybean, are typically planted in Delaware as part of a row crop rotation. Lima beans and peas are also commonly planted in Delaware. In addition to N fixing bacteria, lightning can also convert N2 gas into plant-available forms of N. Also, the combustion of fossil fuel adds N to the atmosphere. When it rains, this source of N can be brought back to earth through atmospheric deposition. Apart from N fixation, the amount of N added to the soil from atmospheric sources is very small compared with the amount added in organic matter and commercial fertilizers.
How Does Nitrogen Behave in the Soil?
Once in the soil, all forms of N can undergo a variety of chemical changes. We already discussed how soil microbes can transform N in plant residues or organic soil amendments into plant available N. This process is called mineralization, and the end product is ammonium. Once in the ammonium form, N can be taken up by plant roots. If ammonium is left on the soil surface, it can be lost to the atmosphere as ammonia gas via a process called volatilization. The risk of ammonia volatilization increases with increasing soil pH. Ammonium can also be converted to nitrate by soil microbes through a process called nitrification. Nitrification typically occurs rapidly (within days) under natural conditions. Use of a urease or nitrification inhibitor can slow nitrification from days to weeks, but ultimately, nitrification will occur.
Like ammonium, soil nitrate can be taken up by plants through the roots. If water percolates down through the soil, nitrate can be moved (leached) down to groundwater. However, if a soil remains waterlogged for a long period of time and loses its oxygen, nitrate can be converted to a gas by microbes and lost to the atmosphere through a process called denitrification.
Nitrogen and Water Quality
Nitrogen can be transported from the soil to surface or groundwater as the field drains following heavy rain or excessive irrigation. When the rainfall or irrigation rate exceeds the soil infiltration capacity, the result is runoff. Runoff can transport soil N, as well as recently applied manure, fertilizers, and plant residues, into lakes, ponds, streams, rivers and bays. In addition, soil nitrate and nitrate fertilizers can be carried downward, or leached, through the soil profile. Once nitrate is leached, it may eventually reach groundwater used for drinking or be discharged by our springs. Leaching is a serious concern in Delaware since a majority of the streams and rivers are groundwater fed.
Once transported, inorganic N can become a water pollutant. For example, nitrate lost in leachate or runoff may contribute to eutrophication of surface water bodies, such as the Delaware In-land Bays and the Chesapeake Bay. Eutrophication is the enrichment of water with nutrients that results in excessive aquatic plant (mostly algae) growth. Over time, oxygen depletion in eutrophic waters can lead to fish kills and the accompanying foul smell. And often, eutrophic water bodies can no longer be used for fishing, swimming, boating or other recreational activities.
Can I Test My Soil for Nitrogen?
Unlike other plant nutrients, most laboratories do not routinely test soil for N because the N cycle is very dynamic. The dynamic nature of the N cycle means that the forms of N in the environment are constantly changing (Figure 1). However, soil test results from a reputable laboratory will include information about N application rates for agronomic, forage, vegetable, and fruit crops commonly grown in Delaware. These recommended rates were determined by scientific research on plant response to N fertilizer rather than on the amount of N measured in a soil sample. It is important to use recommended N fertilizer rates to prevent plant injury and to protect water quality. Even though soils are not tested for N, soil tests are vital because they provide important information about soil pH and the levels of other important plant nutrients (i.e., phosphorus, potassium and magnesium) in the soil. Results from a soil test will help you to properly manage nutrients in your cropping system.
While soil testing is not typically recommended for N management, there are some specialized soil and tissue N tests that are useful in agricultural row crop production. The most common soil N test is the pre-sidedress nitrate test (PSNT) as described in Nitrogen Management for Corn in Delaware: The Pre-Sidedress Nitrate Test (available at http://www.udel.edu/0013390). The PSNT can estimate the contribution of soil N to the overall N requirement of corn in soils with a history of manure application (i.e., manure applied in two or more of the last five years). Managing N based on results of a PSNT can result in economic savings for the grower and reduce the likelihood of groundwater contamination by nitrate-nitrogen. The fall soil nitrate test (FSNT) is a similar N soil test that is useful for determining fall N needs for small grains (Kratochvil & Steinhilber, 2013). In addition, the end-of-season corn stalk nitrate test (CSNT) is used to assess the N status of a corn crop at the end of the growing season as described in End-of-Season Corn Stalk Nitrate Testing to Optimize Nitrogen Management (available at http://www.udel.edu/0013391).
How Can You Help Protect Water Quality
As a grower in Delaware, you can help protect water quality by following best management practices (BMPs) when using manure, commercial fertilizers, or other amendments. We recommend that you follow University of Delaware N rate and timing recommendations, which are available for a variety of crops in the University of Delaware Nutrient Recommendations (available at: http://www.udel.edu/008353) and the Commercial Vegetable Production Recommendations (available at http://www.udel.edu/008353) University of Delaware N recommendations are based on a realistic yield goal as outlined in Estimating Yield Goal for Crops (available at http://www.udel.edu/0013363). For corn, consider split applications of N fertilizer by applying pre-plant manure and starter fertilizers and supplementing in-season with applications of commercial N at sidedress or later in the growing season by fertigation, when possible. When using manure, a PSNT test can guide in-season N application for corn. For soybean and other legumes, skip the manure application and only apply N in-season when expected yields are greater than 70 bu/A as outlined in Nitrogen Management for Soybean (available at http://www.udel.edu/0013393). Finally, consider the FSNT to determine the need for fall fertilization of small grain crops.
References
Kratochvil, R. & Steinhibler, P. (2013). Fall Soil Nitrate Test (FSNT). Extension Brief (EBR-15). University of Maryland Extension. College Park, MD. Available at: https://extension.umd.edu/sites/extension.umd.edu/files/publications/EBR-15%20Fall%20Soil%20Nitrate%20Test.pdf
Shober, A.L. & Reisinger, A.J. (2022). Nitrogen in the Home Landscape. UF-IFAS Publication #SL 254. https://doi.org/10.32473/edis-ss479-2022
About the Author
Amy L. Shober (corresponding author), Professor and Extension Specialist, University of Delaware, Newark, DE (ashober@udel.edu)
About this Publication
Original Publication Date: 2015
Revision date(s): 2025
Based on an original publication produced by A.L. Shober for UF-IFAS (2008).
Peer Reviewer
Richard Taylor, Extension Specialist (retired), Soil Fertility and Crop Management, University of Delaware, Newark, DE
UD Cooperative Extension
This institution is an equal opportunity provider.
In accordance with Federal law and U.S. Department of Agriculture policy, Cooperative Extension is prohibited from discriminating on the basis of race, color, national origin, sex, age, or disability.
