As good as gold
Pyrite nanoparticles from hydrothermal vents rich source of iron
8:31 a.m., May 9, 2011--Similar to humans, the bacteria and tiny plants living in the ocean need iron for energy and growth. But their situation is quite different than ours — for one, they can’t exactly turn to natural iron sources like leafy greens or red meat for a pick-me-up.
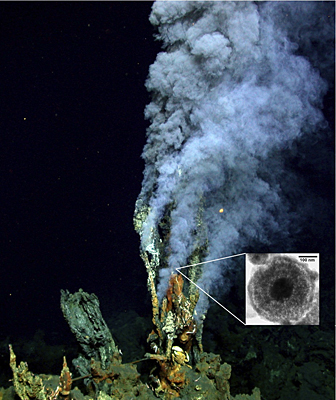
So where does their iron come from? New research published by Nature Geoscience points to a source on the seafloor: minute particles (called nanoparticles) of pyrite, or fool’s gold, from hydrothermal vents at the bottom of the ocean.
Research Stories
Chronic wounds
Prof. Heck's legacy
Scientists already knew the vents’ cloudy plumes emitted from the earth’s interior include pyrite particles, but they thought they were solids that settled back on the ocean bottom. A University of Delaware team has shown that the vents emit a significant amount of pyrite as nanoparticles, which have a diameter that is one thousand times smaller than that of a human hair. Because the nanoparticles are so small, they are dispersed into the ocean rather than falling to the bottom.
Barbara Ransom, program director in the agency that funded the research, the National Science Foundation’s Division of Ocean Sciences, called the discovery "very exciting."
"These particles have long residence times in the ocean and can travel long distances from their sources, forming a potentially important food source for life in the deep sea," she said.
George Luther, project collaborator and professor of oceanography at UD, explained the importance of the pyrite’s lengthy residence times, or how long they exist in their current form. He said the pyrite, which consists of iron and sulfur as iron disulfide, does not rapidly react with oxygen in the seawater to form oxidized iron, or "rust," allowing it to stay intact and move throughout the ocean better than other forms of iron.
"As pyrite travels from the vents to the ocean interior and toward the surface ocean, it oxidizes gradually to release iron, which becomes available in areas where iron is depleted so that organisms can assimilate it, then grow," Luther said. "It’s an ongoing iron supplement for the ocean much as Geritol or multivitamins are for humans."
Growth of the bacteria and tiny plants, known as phytoplankton, can affect atmospheric oxygen and carbon dioxide levels.
Much of the research for the paper, which the journal published on its website on May 8, was completed by alumnus Mustafa Yücel while working on his doctorate with Luther at UD.
The project also received support from Delaware EPSCoR. It involved scientific cruises to the South Pacific and East Pacific Rise using the manned deep-sea submersible Alvin and the remotely operated vehicle Jason, both operated by the Woods Hole Oceanographic Institution.
About the UD research team
George Luther is Maxwell P. and Mildred H. Harrington Professor of Oceanography; Mustafa Yücel earned his doctorate in oceanography from UD in fall 2009 and now holds a post-doctorate position at Benthic Ecogeochemistry Laboratory of France’s Pierre and Marie Curie University (Paris 6) at Banyuls Marine Station; Clara Chan is an assistant professor of geological sciences; and Amy Gartman is an oceanography doctoral student studying with Luther.
Article by Elizabeth Boyle