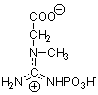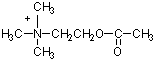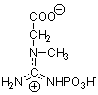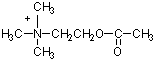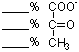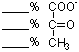CHEM-642 Biochemistry
Name ______________________________
First Hourly Examination
10 AM to noon, Saturday, 17 March 2001
Dr. Hal White - Instructor
There are 10 pages to this examination. Write your name
on each new page. Read every question so that you understand what is being
asked. If you feel any question is unclear or ambiguous, clearly explain
your answer or interpretation. Please call my attention to any errors you
encounter.
You will be given a separate page which has many
metabolic pathways written on it.
This examination will assess your learning, problem-solving
skills, and ability to communicate clearly. It is intended to be challenging
even to the best students in the class. Some of the questions will deal
with material you have not seen before and is not in your text; however,
they can be answered by applying basic principles discussed in the course.
Do not expose your answers to the scrutiny of your neighbors.
Please fold under each page before you go on to the next.
Breakdown of the examination by sections:
Short Answer
10 Points
Structure
20 Points
Problems
46 Points (+5 bonus)
Short Essays
24 Points
Total
100 Points (+5 bonus)
Exam Statistics:
N = 50
Range 22.5 83.5
Mean 56.25 ± 14.71
Part I - Short Answer Questions (1
point each)
____________ 1. Name for two or more proteins that catalyze
the same reaction in a single cell
or organism.
____________ 2. Coenzyme used in reductive reactions
in biosynthesis.
____________ 3. Ferredoxin is a protein containing an
inorganic cluster of these two elements.
____________ 4. An end product of the shikimic acid pathway.
____________ 5. Enzymes activated by ATP are likely to
regulate this class of pathways.
____________ 6. Within an order of magnitude, how long
does the average ATP molecule exist
before it is used?
____________ 7. Inhibitor of prostaglandin synthesis.
____________ 8. The corrin ring of coenzyme B12
and the heme group of cytochromes belong
to this class of molecules.
____________ 9. The cellular location of ketone body
formation.
____________ 10. The prosthetic group of fatty acid synthetase,
4-phospho-pantethiene, is also
part of this coenzyme.
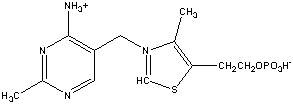
Part II Structures: Identify each compound
and below each provide a brief description of its function (2 points
each).
Draw the indicated structures in the space above each
name. (2 points each)
Oleic acid
Guanosine
Leucine
Galactose
Tryptophan
CMP
Part III Problems
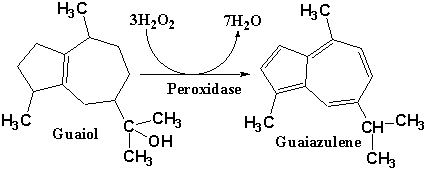
1. Guaiol, a sesquiterpene found in the sap of certain trees, turns
blue when it reacts with peroxides. This reaction was used as a test for
peroxide in the 19th century. Guaiol is derived from farnesyl
pyrophosphate (FPP). In the structure of guaiol below:
A. (3 points) Outline the three
isoprene units that are imbedded in guaiols structure.
B. (2 points) Draw a circle around
the carbon which is derived from the oxygen bearing carbon of FPP.
2. (8 points) Waxes are common in nature.
They provide the water-repellent surfaces to fruits, skin, and feathers
among other things. Waxes are esters of formed from long-chain alcohols
and a fatty acid. Starting with two molecules of palmitic acid,
propose
a biosynthetic pathway for the wax shown below.
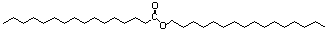
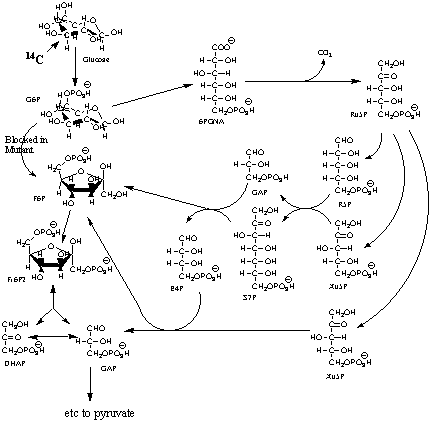
3. Consider the bacteria, Youdea hulliheni,
that normally oxidizes glucose by the Embden-Meyerhof Glycolytic pathway,
but, due to a mutation in the gene for phosphoglucose isomerase (the aldo-keto
isomerase that interconverts G6P and F6P), must use the pentose phosphate
pathway as a detour around the metabolic block as shown below. The cofactors
for the individual enzymes are omitted but you will need to know them.
____ ATP
____ NAD(P) |
-
(4 points) In this modified pathway for glucose oxidation,
five, rather than six, glyceraldehyde-3-Ps (GAP) are produced per three
glucoses. Calculate the net production of ATP and NAD(P)H per glucose in
going from glucose to pyruvate in this pathway. (Use the back of
this page as a work sheet if necessary for this and subsequent questions.)
|
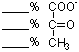 |
B. (6 points) If carbon 3 of glucose were labeled with
14C,
how would the label be distributed in pyruvate?
|
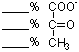 |
C. (3 points) How would the 14C label be
distributed in pyruvate, if the normal glycolytic pathway were operating
alone, i. e. phosphoglucose isomerase is fully functional and the pentose
phosphate pathway were not involved.
|
4. The biosynthesis of phenylalanine
requires erythrose-4-P (E4P) and two moles of phosphoenol pyruvate (PEP)
as precursors as is shown below. Return to Question 3B and the metabolism
of [3-14C] glucose in the bacteria lacking phosphoglucose isomerase.
-
(8 points) Trace the labeled carbons and show which carbons
of phenylalanine would be labeled. (Because the answer here is dependent
on your answer on 3B, I will give up to 6 points for an answer that is
consistent with an incorrect answer to 3B.)
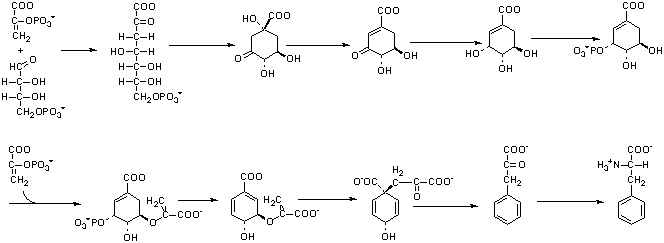
-
Bonus Question (5 points) Do not do this until
you have completed the rest of the examination. What would be the relative
amounts of
14C in each of the labeled carbons of phenylalanine?
5. (12 points) Tryptophan degradation is
a multi-step pathway which leads finally to AcCoA with a
-ketoadipate (a KA) as an intermediate. Fragments
of the last part of this pathway from
a KA to
AcCoA are shown below. Based on analogy to other metabolic pathways and
your biochemical intuition, supply all the missing coenzymes, substrates,
and reactants in the pathway.
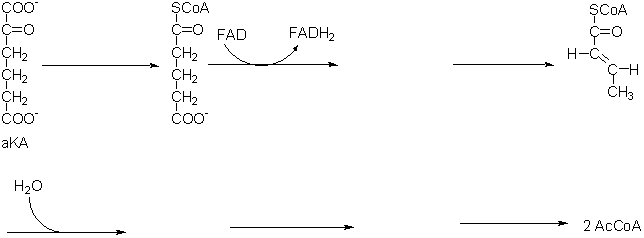
Part IV - Essay Questions (8 Points each) Writing
reflects how you think. Among the "right answers" I will read for the following
questions, some will be better than others because they show greater depth
of understanding, avoid extraneous or inaccurate information, provide a
more logical structure, use appropriate examples, and select words with
precision. Better quality answers will receive higher marks. Therefore,
organize your thoughts before you write.
1. Explain in words and show with clear
examples (illustrated) the concept that despite the fact that NAD and NADP
have similar reduction potentials and chemical properties, enzymes are
specific for either NAD or NADP.
2. By having two copies
of genes encoding each enzyme, diploid organisms like ourselves are cushioned
from the effects of a mutations that generate non-functional enzymes. Interestingly,
genetic carriers for devastating metabolic diseases (persons heterozygous
for a recessive detrimental mutation) produce about half the normal amount
of the affected enzyme and yet rarely show any detrimental effects. Basically
they are normal. In terms of metabolic flux in pathways, how can this be
rationalized?
3. Living organisms do
not violate the laws of thermodynamics. Within terrestrial and shallow
water ecosystems, the energy that maintains the living organisms can be
directly attributed to photosynthetic organisms, the primary producers.
It was a great surprise in the late 1970s when a whole community of organisms
was found in restricted regions of the ocean floor where no light penetrates
[Natl. Geog. 156, 680 (1979)]. The energy input for these organisms apparently
comes from H2S which comes out of nearby hydrothermal springs.
One rather large organism there is Riftia pachyptila, a type of
worm several inches in diameter and up to 10 feet long. These worms lack
a mouth, digestive tract, and anus. Certain parts of their body contain
high densities of endosymbiotic bacteria presumed to be capable of coupling
the aerobic oxidation of sulfide to sulfate to the fixation of CO2
via the Calvin Cycle [Nature 293, 616 (1981), TIBS 7, 201 (1982), and Science
219, 297 (1983)].
Based on your knowledge of metabolism and the information
above, suggest at least two specific pieces of biochemical information
that you would want to know before you would accept the above hypothesis
as possibly valid and not just a science fiction story. Indicate why this
additional information would help your evaluation.
Return to Department's
Home Page, Course
Home Page, Course
Syllabus.
Last updated 21 March 2001
by Hal White.
Copyright 2001, Harold B.
White, Department of Chemistry and Biochemistry, University of Delaware,
Newark, DE 19716