
Publications


Publications from Delaware
(23) Stereospecific Cross Couplings To Set Benzylic, All-Carbon Quaternary Stereocenters in High Enantiopurity
Zhou, Q.; Cobb, K. M.; Tan, T.; Watson, M. P. J. Am. Chem. Soc. 2016, 138(37), 12057–12060.
doi: 10.1021/jacs.6b08075
* Highlighted in Synfacts 2016, 12(11), 1175 *
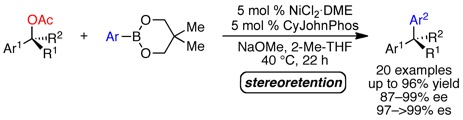
(22) Accessing Both Retention and Inversion Pathways in Stereospecific, Nickel-Catalyzed Miyaura Borylations of Allylic Pivalates
Zhou, Q.; Srinivas, H. D.; Zhang, S.; Watson, M. P. J. Am. Chem. Soc. 2016, 138, 11989–11995.
doi: 10.1021/jacs.6b07396
* Highlighted in Synfacts 2016, 12(11), 1152 *
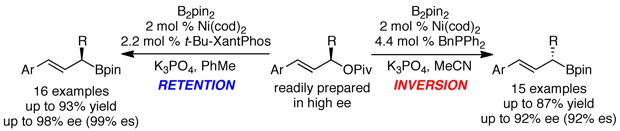
Basch, C. H.; Cobb, K. M.; Watson, M. P. Org. Lett. 2016, 18, 136–139.
doi: 10.1021/acs.orglett.5b03455
Highlighted in Synfacts (Synfacts 2016, 12(4), 0375).
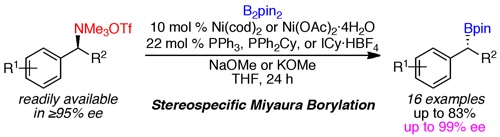
-
(20)Enantioselective Additions of Copper Acetylides to Cyclic Iminium and Oxocarbenium Ions
Liu, J.; Dasgupta, S.; Watson, M. P. Beilstein J. Org. Chem. 2015, 11, 2696–2706.
doi: 10.3762/bjoc.11.290
-
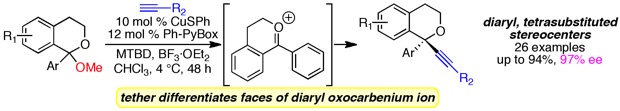
(19)Enantioselective, Copper(I)-Catalyzed Alkynylation of Oxocarbenium Ions to Set Diaryl Tetrasubstituted Stereocenters
Dasgupta, S.; Rivas, T.; Watson, M. P. Angew. Chem. 2015, 54, 14154–14158.
doi: 10.1002/anie/201507373
Highlighted in Synfacts (Synfacts 2016, 12(2), 0169).
-
(18)Stereoselective Synthesis of Trisubstituted Vinyl Bromides via Addition of Alkynes to Oxocarbenium Ions
Ehle, A. R.; Morris, M. G.; Klebon, B. D.; Yap, G. P. A.; Watson, M. P. Synlett 2015, 26(19), 2702–2706.
doi: 10.1055/s-0035-1560265
(Invited contribution to special issue highlighting 2015 EuCheMS Young Investigator Workshop participants)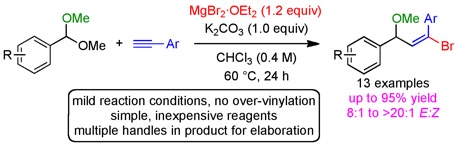
-
(17) Diastereoselective, Zinc-Catalyzed Alkynylation of alpha-Bromo Oxocarbenium Ions
Haidzinskaya, T.; Kerchner, H. A.; Liu, J.; Watson, M. P. Org. Lett. 2015, 17(15), 3857–3859.
doi: 10.1021/acs.orglett.5b01838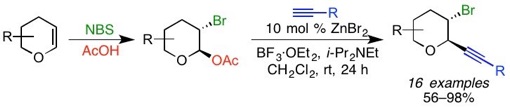
-
(16) Enantioselective Copper-Catalyzed Alkynylation of Benzopyranyl Oxocarbenium Ions
Srinvias, H. D.; Maity, P.; Yap, G. P. A.; Watson, M. P. J. Org. Chem. 2015, 80(8), 4003–4016.
doi: 10.1021/acs.job.5b00364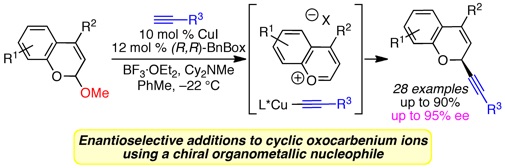
-
(15)Enantiospecific, Nickel-Catalyzed Cross-Couplings of Allylic Pivalates and Arylboroxines
Srinivas, H. D.; Zhou, Q.; Watson, M. P. Org. Lett. 2014, 16(13), 3596–3599.
doi: 10.1021/ol5016724
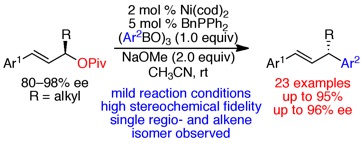
-
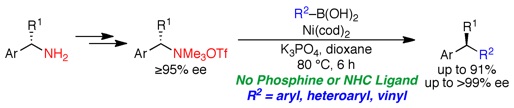
(14) A General, Simple Catalyst for Enantiospecific Cross Couplings of Benzylic Ammonium Triflates and Boronic Acids: No Phosphine Ligand Required
Shacklady-McAtee, D. M.; Roberts, K. M.; Basch, C. H.; Song, Y.-G.; Watson, M. P. Tetrahedron 2014, 70(27–28), 4257–4263.
doi: 10.1016/j.tet.2014.03.039
Invited contribution to Symposium in Print in Honor of Prof. Sarah Reisman’s Tetrahedron Young Investigator Award – Congratulations, Sarah!
Highlighted in Synfacts (Synfacts 2014, 10, 1060).
-
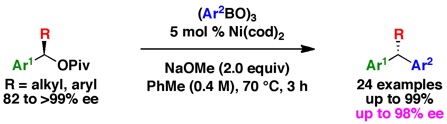
(13) Nickel-Catalyzed Cross-Couplings of Benzylic Pivalates with Arylboroxines: Stereospecific Formation of Diarylalkanes and Triarylmethanes
Zhou, Q.; Srinivas, H. D.; Dasgupta, S.; Watson, M. P. J. Am. Chem. Soc. 2013, 135(9), 3307–3310.
doi: 10.1021/ja312087x
*Highlighted in Synfacts (Synfacts 2013, 9(6), 646)*
-
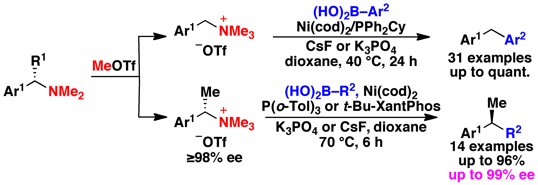
(12)NIckel-Catalyzed Cross Couplings of Benzylic Ammonium Salts and Boronic Acids: Stereospecific Formation of Diarylethanes via C–N Bond Activation
Maity, P.; Shacklady-McAtee, D. M.; Yap, G. P. A.; Sirianni, E. R.; Watson, M. P. J. Am. Chem. Soc. 2013, 135(1), 280–285.
doi: 10.1021/ja3089422
-
(11)Controlling Enantioselectivity in Additions to Cyclic Oxocarbenium Ions via Transition-Metal Catalysis (SynPacts article)
Watson, M. P.; Maity, P. Synlett 2012, 23(12), 1705–1708.
doi: 10.1055/s-0031-1290697
-
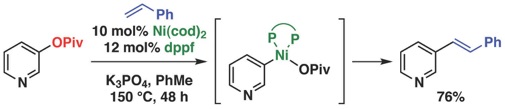
(10)Nickel(0)-Catalyzed Heck Cross-Couplings via Activation of Aryl C–OPiv Bonds
Ehle, A. R.; Zhou, Q.; Watson, M. P. Org. Lett. 2012, 14(5), 1202–1205.
doi: 10.1021/ol203322v
*One of the top 10 most read articles in Organic Letters from Jan–Mar 2012*
-
(9) Copper-Catalyzed Enantioselective Additions to Oxocarbenium Ions: Alkynylation of Isochroman Acetal
Maity, P.; Srinivas, H. D.; Watson, M. P. J. Am. Chem. Soc. 2011, 133(43), 17142–17145.
doi: 10.1021/ja207585p
*Highlighted in Synfacts (Synfacts 2012, 8, 78) and as a SynStory (Dec 2011)*
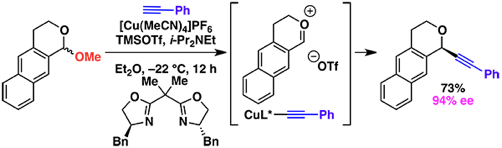
-
(8)Nickel(0)-Catalyzed Cyclization of N-Benzoylaminals for Isoindolinone Synthesis
Shacklady-McAtee, D. M.; Dasgupta, S.; Watson, M. P. Org. Lett. 2011, 13(13), 3490–3493.
doi: 10.1021/ol201248c
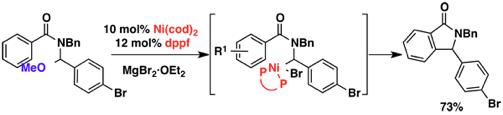
Publications from Supervised Career
-
(7)Asymmetric Intramolecular Arylcyanation of Unactivated Olefins via C–CN Bond Activation
Watson, M. P.; Jacobsen, E. N. J. Am. Chem. Soc. 2008, 130(38), 12594–12595.
-
(6)Kinetic and Computational Analysis of the Palladium(II)-Catalyzed Asymmetric Allylic Trichloroacetimidate Rearrangement: Development of a Model for Enantioselectivity
Watson, M. P.; Overman, L. E.; Bergman, R. G. J. Am. Chem. Soc. 2007, 129(16), 5031–5044.
-
(5)Preparation of the COP Catalysts: [(S)-COP-OAc]2, [(S)-COP-Cl]2, and (S)-COP-hfacac
Anderson, C. E.; Kirsch, S. F.; Overman, L. E.; Richards, C. J.; Watson, M. P. Org. Synth. 2007, 84, 148–155.
-
(4)Preparation of (eta5-(S)-2-(4-methylethyl)oxazolinylcyclopentadienyl)-(eta4-tetraphenylcyclobutadiene)cobalt
Anderson, C. E.; Overman, L. E.; Richards, C. J.; Watson, M. P.; White, N. Org. Synth. 2007, 84, 139–147.
-
(3)Asymmetric Rearrangement of Allylic Trichloroacetimidates: Preparation of (S)-2,2,2,-Trichloro-N-(1-propylallyl)acetamide
Anderson, C. E.; Overman, L. E.; Watson, M. P. Org. Synth. 2005, 82, 134–139.
-
(2)Monomeric Cobalt Oxazoline Palladacycles (COP). Useful Catalysis for Catalytic Asymmetric Rearrangement of Allylic Trichloroacetimidates
Kirsch, S. F.; Overman, L. E.; Watson, M. P. J. Org. Chem. 2004, 69(23), 8101–8104.
-
(1)Catalytic Asymmetric Rearrangement of Allylic N-Aryl Trifluoroacetimidates. A Useful Method for Transforming Prochiral Allylic Alcohols to Chiral Allylic Amines
Overman, L. E.; Owen, C. E.; Pavan, M. M.; Richards, C. J. Org. Lett. 2003, 5(11), 1809–1812.